|
Your objective is to:
Recognize sublimation, deposition and triple point from a microscopic point of view.
You will be able to:
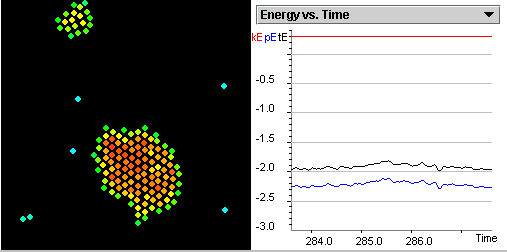
Describe the process of deposition and explain it in terms
of potential energy.
Describe the process of sublimation and explain it in terms
of energy.
Compare and explain the potential energies of particles that
exist at the triple point.
Describe molecular motion at the triple point.
|