|
Your objective is to:
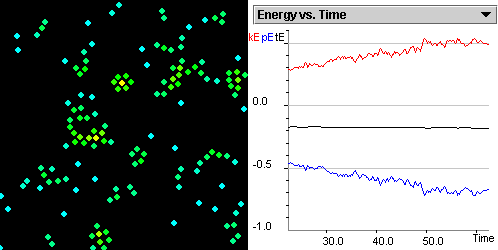
Recognize the difference
between a gas and a liquid in terms of the energies of the particles.
You will be able to:
Differentiate among the liquid and gas state in terms of
average kinetic energy and the average potential energy of the
particles.
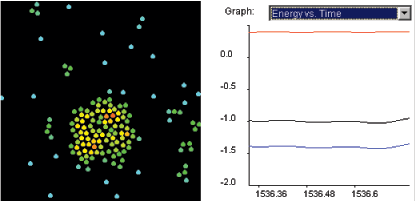
Contrast the potential energy of a molecule in the center
of a droplet with that on the edge of the droplet.
State the relationship between the trajectory of a particle
and its potential energy.
Explain the release of latent heat when a gas
condenses.
|