|
Your objective is to:
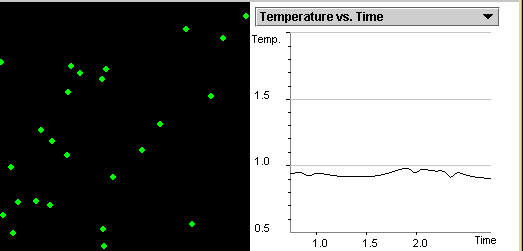
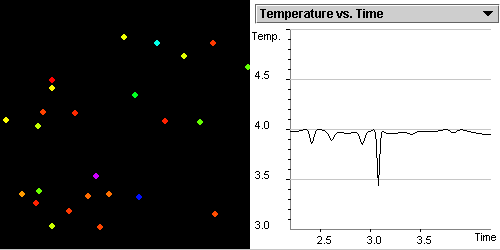
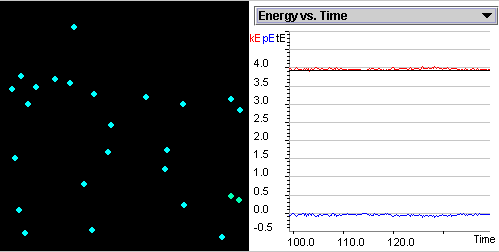
Investigate the correlation
between the kinetic,
potential energies and temperature as gas particles move and collide.
You will be able to:
Define potential energy and kinetic energy and give an
example of each.
Discuss the relationship among potential energy, kinetic
energy, and total energy before, during, and after a two-particle
collision at a given temperature.
State the relationship between the average kinetic energy
of randomly moving particles and the temperature of the system.
Describe what happen to the speed of particles in a system
as the temperature is raised.
|