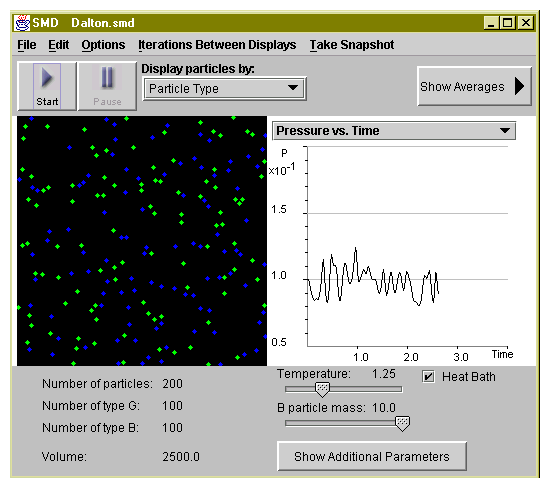
In our simulation it is more convenient to use number of particles instead of number of moles. Moreover, in the simulation units the Boltzmann constant k=1. Hence, the partial pressures
|
where Ni is just a number of molecules of certain type.
|
| N/V | P | T | |
| 100 Blue and 100 Green | |||
| 100 Blue | |||
| 100 Green |
|
|
|
|
 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|